Assessment of the Capillary Electrophoresis Study
The Objective of the Study
Our client is a leading global and consulting company who wanted to get opinions and attitudes about issues related to Capillary Electrophoresis (CE) analysis software
Key Screening Points
- Experts who are familiar with capillary electrophoresis in fragment analysis or Sanger sequencing, and the process of analysis and reporting at their laboratory.
- Experts are familiar with lab’s budgeting and pricing decisions for capillary electrophoresis in genetic analysis applications.
- Experts involved in Academic research institution, Commercial service provider, Academic medical centre, Large or regional hospital network (i.e., more than 500 beds) Integrated Delivery Network Health System (IDN), Community-based hospital (i.e., fewer than 500 beds), Biopharmaceutical or Contract Research Organization (CRO).
- Experts should be working in Pre-clinical, basic and/or translational research lab, Clinical research lab, Clinical testing lab, Commercial clinical specialty laboratory or Commercial clinical reference laboratory.
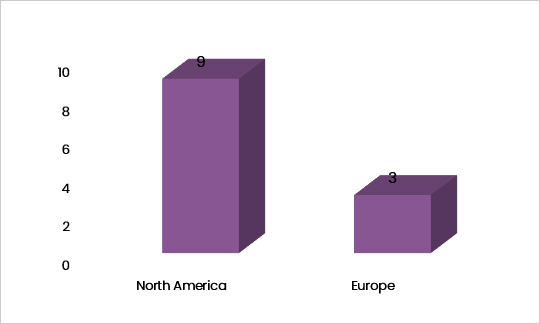
- The geographic scope was USA, Canada, UK and Ireland.